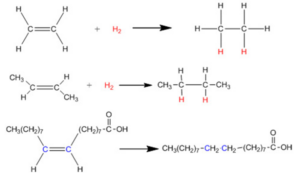
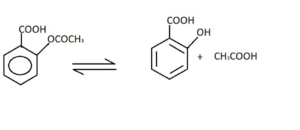
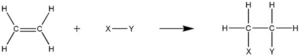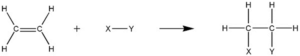Introduction to Exchange Reaction
An exchange reaction is a type of chemical reaction that involves the exchange of ions between reactants, resulting in the formation of new products. This process is also known as ion exchange. In an exchange reaction, the ions present in the reactants are rearranged to form new combinations, leading to the formation of different compounds.
Exchange reactions play a crucial role in various fields, including chemistry, biology, and environmental science. They occur in both natural and synthetic systems and have significant implications for understanding the behavior of substances in different environments.
Let’s explore the definition, characteristics, and importance of exchange reactions in more detail.
Definition of Exchange Reaction
An exchange reaction can be defined as a chemical reaction in which ions are exchanged between reactants, resulting in the formation of new compounds. This process occurs due to the differences in the chemical properties of the ions involved.
In an exchange reaction, the reactants consist of ions that are either positively charged (cations) or negatively charged (anions). These ions can be present in a variety of chemical compounds, such as salts, acids, or bases. When the reactants come into contact, the ions are rearranged, leading to the formation of new compounds with different combinations of ions.
Characteristics of Exchange Reaction
Exchange reactions exhibit several distinct characteristics that differentiate them from other types of chemical reactions:
-
Reversible Reaction: Exchange reactions are reversible, meaning they can proceed in both the forward and backward directions. This reversibility is due to the dynamic nature of the exchange process, where ions continuously interact and exchange positions.
-
Equilibrium: Like other reversible reactions, exchange reactions reach a state of equilibrium when the rates of the forward and backward reactions become equal. At equilibrium, the concentrations of the reactants and products remain constant, although the exchange process continues.
-
Rate of Reaction: The rate at which an exchange reaction occurs depends on various factors, including the nature of the reactants, temperature, and concentration. The presence of a catalyst can also influence the rate of the reaction by providing an alternative reaction pathway with lower activation energy.
-
Reaction Mechanism: Exchange reactions follow a specific reaction mechanism, which describes the step-by-step process by which reactants transform into products. Understanding the reaction mechanism is crucial for predicting and controlling the outcome of an exchange reaction.
-
Stoichiometry: The stoichiometry of an exchange reaction refers to the quantitative relationship between the reactants and products. It is determined by the balanced chemical equation, which indicates the ratio of ions exchanged during the reaction.
-
Reaction Kinetics: The study of exchange reactions also involves investigating the kinetics of the reaction, which focuses on the rate at which the reaction proceeds. Reaction kinetics provides insights into the factors that influence the speed and efficiency of the exchange process.
Importance of Exchange Reaction in Chemistry
Exchange reactions have significant importance in the field of chemistry. They serve as fundamental building blocks for understanding various chemical processes and phenomena. Here are some key reasons why exchange reactions are important:
-
Ion Exchange: Exchange reactions are widely used in ion exchange processes, where ions are selectively exchanged between a solid material (such as a resin) and a liquid solution. This technique is employed in water treatment, purification, and separation processes.
-
Environmental Applications: Exchange reactions play a crucial role in environmental science, particularly in soil chemistry. They influence the availability and mobility of nutrients and contaminants in soil, affecting plant growth and environmental remediation efforts.
-
Material Synthesis: Exchange reactions are utilized in the synthesis of various materials, such as nanocrystals and surfactants. By controlling the exchange of ions, researchers can manipulate the structure, form, and morphology of these materials, leading to unique properties and applications.
-
Chemical Analysis: Exchange reactions are employed in analytical chemistry for the determination of ions in a sample. Techniques such as ion chromatography and ion-selective electrodes rely on the principles of exchange reactions to quantify the concentration of specific ions.
In conclusion, exchange reactions are an essential aspect of chemistry, with wide-ranging applications in various fields. Understanding the definition, characteristics, and importance of exchange reactions provides a foundation for exploring their role in chemical processes and their impact on the natural and synthetic world.
Precipitation Reaction Examples
Precipitation reactions are a type of chemical reaction that occur when two aqueous solutions are mixed together, resulting in the formation of an insoluble solid called a precipitate. These reactions are commonly observed in various fields, including chemistry, biology, and environmental science. In this section, we will explore two examples of precipitation reactions and their outcomes.
Precipitation Reaction between Sodium Chloride and Silver Nitrate
One common example of a precipitation reaction is the reaction between sodium chloride (NaCl) and silver nitrate (AgNO3). When these two solutions are combined, a white precipitate of silver chloride (AgCl) is formed. This reaction can be represented by the following equation:
NaCl(aq) + AgNO3(aq) → AgCl(s) + NaNO3(aq)
In this reaction, the sodium chloride and silver nitrate ions exchange partners, resulting in the formation of the insoluble silver chloride precipitate. The sodium and nitrate ions remain in solution as sodium nitrate.
Precipitation Reaction between Calcium Chloride and Potassium Hydroxide
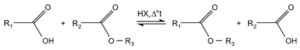
Another example of a precipitation reaction involves the reaction between calcium chloride (CaCl2) and potassium hydroxide (KOH). When these two solutions are mixed, a white precipitate of calcium hydroxide (Ca(OH)2) is formed. The balanced chemical equation for this reaction is as follows:
CaCl2(aq) + 2KOH(aq) → Ca(OH)2(s) + 2KCl(aq)
In this reaction, the calcium chloride and potassium hydroxide ions exchange partners, resulting in the formation of the insoluble calcium hydroxide precipitate. The potassium and chloride ions remain in solution as potassium chloride.
Precipitation reactions are often used in laboratory settings to identify the presence of specific ions in a solution. By observing the formation of a precipitate, scientists can determine the identity of the ions present. These reactions also have practical applications in industries such as wastewater treatment, where they are used to remove unwanted ions from solution.
In summary, precipitation reactions occur when two aqueous solutions are mixed, resulting in the formation of an insoluble solid called a precipitate. The examples discussed above illustrate how different combinations of ions can lead to the formation of specific precipitates. These reactions play a crucial role in various scientific and industrial processes, making them an important area of study in chemistry.
Neutralization Reaction Example
Neutralization reactions are a type of chemical reaction that occur when an acid and a base react with each other to form a salt and water. These reactions are important in various fields, including chemistry, biology, and environmental science. In this section, we will explore a specific example of a neutralization reaction between hydrochloric acid (HCl) and potassium hydroxide (KOH).
Neutralization Reaction between Hydrochloric Acid and Potassium Hydroxide
When hydrochloric acid and potassium hydroxide are combined, a neutralization reaction occurs. The reaction can be represented by the following balanced chemical equation:
HCl + KOH → KCl + H2O
In this reaction, hydrochloric acid (HCl) is an acid, and potassium hydroxide (KOH) is a base. The reaction between the two results in the formation of potassium chloride (KCl) and water (H2O).
During the reaction, the hydrogen ion (H+) from the acid combines with the hydroxide ion (OH-) from the base to form water. The remaining ions, potassium (K+) from the base and chloride (Cl-) from the acid, combine to form the salt potassium chloride.
Importance of Neutralization Reactions
Neutralization reactions have several important applications. One of the most common uses is in the field of medicine. Antacids, for example, are medications that contain bases to neutralize excess stomach acid. By neutralizing the acid, antacids help relieve symptoms of heartburn and indigestion.
Neutralization reactions also play a crucial role in wastewater treatment. In this process, acidic or basic wastewater is treated with a neutralizing agent to bring the pH to a more neutral level. This helps to prevent environmental damage and ensure the safety of aquatic life.
Understanding the Reaction Process
To understand the neutralization reaction between hydrochloric acid and potassium hydroxide, it is important to consider the reaction mechanism. The reaction proceeds in a stepwise manner, involving the transfer of protons (H+) and hydroxide ions (OH-).
-
Proton transfer: The hydrogen ion (H+) from the hydrochloric acid donates a proton to the hydroxide ion (OH-) from the potassium hydroxide. This proton transfer results in the formation of water.
-
Ion exchange: The remaining ions, potassium (K+) from the base and chloride (Cl-) from the acid, combine to form the salt potassium chloride.
The reaction is reversible, meaning it can proceed in both the forward and backward directions. However, in most cases, the reaction is driven towards the formation of the products (KCl and H2O) due to the strong acid-base interaction.
Conclusion
In this section, we explored a specific example of a neutralization reaction between hydrochloric acid and potassium hydroxide. We discussed the balanced chemical equation, the importance of neutralization reactions in various fields, and the reaction process. Neutralization reactions are essential in many aspects of our daily lives, from medicine to environmental protection. Understanding these reactions helps us comprehend the underlying chemistry and their practical applications.
Gas Evolution Reaction Examples
Gas evolution reactions are chemical reactions that result in the formation of a gas as one of the products. These reactions are commonly encountered in various fields, including chemistry, biology, and industry. In this section, we will explore two examples of gas evolution reactions and discuss their underlying principles and applications.
Gas Evolution Reaction between Zinc and Concentrated Sulfuric Acid
One example of a gas evolution reaction is the reaction between zinc and concentrated sulfuric acid. When zinc is added to concentrated sulfuric acid, hydrogen gas is evolved. This reaction can be represented by the following equation:
Zn + H2SO4 → ZnSO4 + H2↑
In this reaction, zinc (Zn) reacts with sulfuric acid (H2SO4) to form zinc sulfate (ZnSO4) and hydrogen gas (H2) as a product. The upward arrow (↑) indicates the evolution of gas.
The gas evolution reaction between zinc and concentrated sulfuric acid is a classic example of a redox reaction. Zinc undergoes oxidation, losing electrons to form zinc ions (Zn2+), while hydrogen ions (H+) from sulfuric acid are reduced, gaining electrons to form hydrogen gas. This reaction is exothermic, meaning it releases heat energy.
This reaction has several applications. One of the most common uses is in the production of hydrogen gas. Hydrogen gas is widely used in various industries, including the production of ammonia, petroleum refining, and as a fuel source for fuel cells. The gas evolution reaction between zinc and sulfuric acid provides a convenient and efficient method for generating hydrogen gas.
Gas Evolution Reaction between Sodium Sulfide and Hydrochloric Acid
Another example of a gas evolution reaction is the reaction between sodium sulfide and hydrochloric acid. When sodium sulfide is added to hydrochloric acid, hydrogen sulfide gas is evolved. The reaction can be represented by the following equation:
Na2S + 2HCl → 2NaCl + H2S↑
In this reaction, sodium sulfide (Na2S) reacts with hydrochloric acid (HCl) to form sodium chloride (NaCl) and hydrogen sulfide gas (H2S) as a product. Once again, the upward arrow (↑) indicates the evolution of gas.
The gas evolution reaction between sodium sulfide and hydrochloric acid is an example of an acid-base reaction. Sodium sulfide, a base, reacts with hydrochloric acid, an acid, to form sodium chloride, a salt, and hydrogen sulfide gas. Hydrogen sulfide gas is known for its distinctive odor, resembling that of rotten eggs.
This reaction has various applications. Hydrogen sulfide gas is used in the production of sulfuric acid, as a reducing agent in chemical processes, and in the synthesis of various organic compounds. Additionally, hydrogen sulfide gas is utilized in analytical chemistry for the detection and identification of metal ions.
In conclusion, gas evolution reactions play a significant role in various chemical processes and applications. The examples discussed, the reaction between zinc and concentrated sulfuric acid, and the reaction between sodium sulfide and hydrochloric acid, demonstrate the formation of hydrogen gas and hydrogen sulfide gas, respectively. These reactions showcase the diverse nature of gas evolution reactions and their importance in both industrial and laboratory settings.
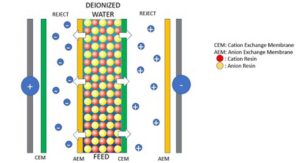
Ion Exchange Reaction Example
In the realm of chemistry, ion exchange reactions play a crucial role in various processes. These reactions involve the exchange of ions between two substances, resulting in the formation of new compounds. Let’s explore an example of an ion exchange reaction to gain a better understanding of this fascinating phenomenon.
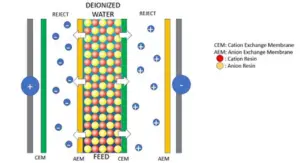
Ion Exchange Resin
One common application of ion exchange reactions is in the use of ion exchange resins. These resins are porous materials that possess the ability to exchange ions with the surrounding solution. They are widely used in water treatment, purification processes, and even in the pharmaceutical industry.
Ion exchange resins consist of a three-dimensional network structure with numerous tiny pores. These pores act as sites for the exchange of ions. The resin is typically made up of organic polymers, which can be modified to have specific properties depending on the desired application.
Example of an Ion Exchange Reaction
To illustrate an ion exchange reaction, let’s consider the removal of potassium ions from soil using an ion exchange resin. In this process, the resin is in the form of small beads or granules packed in a column. The soil sample is passed through the column, allowing the exchange of ions to take place.
Initially, the resin contains sodium ions, while the soil contains potassium ions. As the soil passes through the resin column, the potassium ions in the soil are attracted to the resin’s surface due to the resin’s affinity for potassium. At the same time, the sodium ions on the resin are released into the soil.
The ion exchange reaction can be represented by the following equation:
Soil (K+) + Resin (Na+) ⟶ Soil (Na+) + Resin (K+)
As a result of this reaction, the potassium ions are effectively removed from the soil, and the resin becomes loaded with potassium ions. This process can be repeated multiple times until the resin is saturated with potassium ions and can no longer exchange them with the soil.
Benefits and Applications
Ion exchange reactions have numerous benefits and applications. Some of the advantages of using ion exchange resins include:
-
Water purification: Ion exchange resins are widely used in water treatment processes to remove impurities such as heavy metals, nitrates, and organic compounds.
-
Softening hard water: Ion exchange resins can be employed to remove calcium and magnesium ions from hard water, preventing the formation of scale in pipes and appliances.
-
Pharmaceutical industry: Ion exchange resins are utilized in the purification and separation of pharmaceutical compounds, ensuring the production of high-quality drugs.
-
Nuclear industry: Ion exchange resins play a crucial role in the treatment and purification of radioactive waste in the nuclear industry.
In conclusion, ion exchange reactions are fundamental processes that occur in various fields, ranging from water treatment to pharmaceutical production. Understanding these reactions and their applications allows scientists and engineers to develop innovative solutions for a wide range of challenges. The example of an ion exchange reaction involving the removal of potassium ions from soil using an ion exchange resin demonstrates the practicality and effectiveness of this process.
Hydrogen Isotope Exchange Reaction Example
In chemistry, exchange reactions play a crucial role in understanding the behavior of various compounds and elements. One such example is the hydrogen isotope exchange reaction. This reaction involves the exchange of hydrogen isotopes, such as deuterium (^2H) or tritium (^3H), with hydrogen atoms (^1H) in a molecule or compound. Let’s explore this fascinating reaction in more detail.
Hydrogen Isotope Exchange Reaction
The hydrogen isotope exchange reaction is a chemical process where hydrogen atoms in a molecule or compound are replaced by hydrogen isotopes. This exchange can occur between different hydrogen isotopes or between hydrogen isotopes and hydrogen atoms. The reaction is typically reversible, meaning that the exchange can occur in both the forward and backward directions.
Understanding the Process
To better understand the hydrogen isotope exchange reaction, let’s consider an example involving the exchange of deuterium (^2H) with hydrogen atoms (^1H) in a compound. This reaction can be represented by the following equation:
Compound with ^1H + ^2H2O ⇌ Compound with ^2H + H2O
In this example, the compound initially contains hydrogen atoms (^1H). When it comes into contact with deuterium oxide (^2H2O), the hydrogen atoms in the compound can undergo an exchange with the deuterium atoms in the water molecule. As a result, the compound now contains deuterium atoms (^2H) instead of hydrogen atoms (^1H).
Importance and Applications
The hydrogen isotope exchange reaction has significant importance in various fields of study. It is widely used in research and industrial applications to study the behavior of molecules, determine reaction mechanisms, and investigate the structure of compounds. This reaction also finds applications in fields such as pharmaceuticals, environmental science, and materials science.
In pharmaceutical research, hydrogen isotope exchange reactions help in studying the metabolism of drugs and understanding their interactions with biological systems. This information is crucial for developing safe and effective medications.
In environmental science, hydrogen isotope exchange reactions are used to trace the movement of water in natural systems. By analyzing the isotopic composition of water samples, scientists can gain insights into the origin, transport, and mixing of water in rivers, lakes, and groundwater.
In materials science, hydrogen isotope exchange reactions are employed to modify the properties of materials. By selectively replacing hydrogen atoms with deuterium atoms, researchers can alter the structure and behavior of materials, leading to improved performance in various applications.
Conclusion
The hydrogen isotope exchange reaction is a fascinating chemical process that involves the exchange of hydrogen atoms with hydrogen isotopes. This reaction has diverse applications and plays a crucial role in understanding the behavior of compounds and materials. By studying this reaction, scientists can gain valuable insights into the structure, reactivity, and properties of substances, leading to advancements in various fields of science and technology.
Metal Halogen Exchange Reaction Example
The metal halogen exchange reaction is a fascinating chemical process that involves the exchange of halogen ions between metal ions in a solution. This reaction is commonly observed in various fields, including soil chemistry, materials science, and nanotechnology. Let’s explore an example of a metal halogen exchange reaction to understand its significance and implications.
Metal Halogen Exchange Reaction in Soil Chemistry
In soil chemistry, metal halogen exchange reactions play a crucial role in determining the availability and mobility of essential nutrients for plants. One such example is the exchange of potassium ions (K+) with chloride ions (Cl-) in the soil. This exchange reaction occurs in the soil pore structure, facilitated by the presence of surfactants.
Surfactants, which are commonly found in soil, act as catalysts in this exchange process. They enhance the rate of the reaction and promote the transfer of ions between the soil particles. The stoichiometry of the reaction depends on the specific surfactant and the concentration of the ions involved.
Metal Halogen Exchange Reaction in Materials Science
In materials science, metal halogen exchange reactions are utilized to modify the structure and properties of materials. For instance, researchers have explored the exchange of halogen ions with metal ions in nanocrystals to control their morphology and composition. This exchange reaction allows for the formation of nanocrystals with specific structures and desired properties.
By carefully selecting the metal ions and the halogen ions involved in the exchange, scientists can tailor the properties of the nanocrystals, such as their size, shape, and optical properties. This control over the nanocrystal structure opens up new possibilities for applications in fields like electronics, photonics, and catalysis.
Metal Halogen Exchange Reaction in Nanotechnology
In nanotechnology, metal halogen exchange reactions are employed to fabricate nanomaterials with unique properties. An example of such a reaction is the exchange of anions and cations in surfactant-templated nanocrystals. The surfactant molecules act as templates, guiding the formation of nanocrystals with specific structures.
During the exchange reaction, the anions and cations in the surfactant-templated nanocrystals are replaced with different ions, resulting in a transformation of the nanocrystal structure. This process allows for the formation of nanomaterials with tailored properties, such as enhanced conductivity, improved stability, or specific surface functionalities.
In conclusion, metal halogen exchange reactions are versatile and powerful tools in various scientific disciplines. They enable the manipulation of chemical structures and properties, leading to advancements in fields like soil chemistry, materials science, and nanotechnology. By understanding and harnessing these exchange reactions, researchers can unlock new possibilities for the development of innovative materials and technologies.
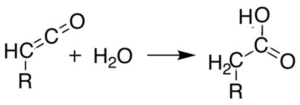
Can You Explain the Acetic Acid Lewis Structure in Detail?
The acetic acid lewis structure explained reveals the arrangement of atoms and their bonding in acetic acid. It consists of two carbon atoms, four hydrogen atoms, and two oxygen atoms. The central carbon atom is bonded to two oxygen atoms, one hydrogen atom, and another carbon atom. This structure helps us understand the chemical properties and reactions of acetic acid.
Frequently Asked Questions (FAQ)
When Does an Exchange Reaction Take Place?
An exchange reaction occurs when there is a chemical reaction between two or more reactants, resulting in the formation of new products. This type of reaction involves the exchange of ions or groups between the reactants, leading to a rearrangement of the molecular structure.
Exchange reactions commonly take place in various fields, including chemistry, biology, and environmental science. In chemistry, exchange reactions are often observed when different compounds react with each other, resulting in the formation of new compounds. For example, when a metal reacts with an acid, an exchange reaction occurs, leading to the formation of a salt and the release of hydrogen gas.
In biological systems, exchange reactions play a crucial role in various processes. For instance, during cellular respiration, the exchange of electrons and protons takes place in the mitochondria, resulting in the production of ATP, the energy currency of cells.
In environmental science, exchange reactions are particularly important in soil chemistry. Soil is composed of various minerals and organic matter, and exchange reactions occur between the soil particles and the surrounding solution. These reactions involve the exchange of ions between the soil particles and the solution, influencing the availability of nutrients for plants.
Is an Exchange Reaction Reversible?
Yes, an exchange reaction can be reversible. In a reversible reaction, the reactants can form products, and the products can also react to form the original reactants. This means that the reaction can proceed in both the forward and backward directions.
The reversibility of an exchange reaction depends on several factors, including the reaction conditions, the presence of a catalyst, and the stoichiometry of the reaction. If the reaction is reversible, it will reach a state of equilibrium, where the rate of the forward reaction is equal to the rate of the backward reaction.
For example, consider the reaction between potassium chloride (KCl) and silver nitrate (AgNO3) to form silver chloride (AgCl) and potassium nitrate (KNO3). Initially, the forward reaction will occur, resulting in the formation of AgCl and KNO3. However, as the reaction progresses, the backward reaction will also take place, leading to the reformation of KCl and AgNO3. At equilibrium, the concentrations of the reactants and products will remain constant, indicating a reversible exchange reaction.
What is the Difference between an Addition Reaction and an Exchange Reaction?
While both addition reactions and exchange reactions involve the formation of new products, there are significant differences between the two.
An addition reaction occurs when two or more reactants combine to form a single product. In this type of reaction, the reactants add to each other without any exchange of atoms or groups. The reaction typically involves the breaking of multiple bonds and the formation of new bonds. Addition reactions are commonly observed in organic chemistry, where unsaturated compounds react with other molecules to form saturated compounds.
On the other hand, an exchange reaction involves the exchange of atoms or groups between the reactants, resulting in the formation of new compounds. This type of reaction often occurs when there is a transfer of ions or functional groups between the reactants. Exchange reactions can occur in various systems, including chemical, biological, and environmental systems.
In summary, the main difference between addition reactions and exchange reactions lies in the nature of the chemical transformation. Addition reactions involve the combination of reactants to form a single product, while exchange reactions involve the exchange of atoms or groups between the reactants, leading to the formation of new compounds.
Conclusion
In conclusion, exchange reactions are a fundamental concept in chemistry that involve the transfer of atoms, ions, or groups between different molecules. These reactions play a crucial role in various chemical processes, such as acid-base reactions, redox reactions, and precipitation reactions. Through the exchange of species, new compounds are formed, and the reaction proceeds towards equilibrium. Exchange reactions can be observed in everyday life, such as the rusting of iron, the neutralization of acids with bases, and the formation of salts. Understanding exchange reactions is essential for comprehending the behavior of substances in chemical reactions and their impact on the world around us. By studying and analyzing exchange reactions, scientists can gain insights into the underlying principles of chemistry and apply this knowledge to various fields, including medicine, environmental science, and materials science.
Frequently Asked Questions
What is an exchange reaction example?
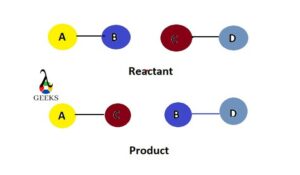
An exchange reaction example involves the interchange of atoms or groups between two or more compounds. For instance, the reaction between sodium chloride (NaCl) and silver nitrate (AgNO3) to form sodium nitrate (NaNO3) and silver chloride (AgCl) is an example of an exchange reaction.
What is an exchange reaction in chemistry?
In chemistry, an exchange reaction refers to a chemical reaction where atoms or groups are interchanged between different compounds. This type of reaction involves the breaking and formation of chemical bonds. It is also known as a double displacement or metathesis reaction.
Can you provide an example of a halogen exchange reaction?
Certainly! An example of a halogen exchange reaction is the reaction between chlorine (Cl2) and potassium iodide (KI) to form potassium chloride (KCl) and iodine (I2). This reaction involves the exchange of halogen atoms between the compounds.
Is there an exchange reaction example in the body?
Yes, there are exchange reactions that occur in the body. One example is the exchange of oxygen and carbon dioxide in the lungs during respiration. Oxygen is taken up by red blood cells, while carbon dioxide is released from the cells and exchanged with oxygen in the alveoli of the lungs.
How would you define an exchange reaction with an example?
An exchange reaction, also known as a double displacement or metathesis reaction, involves the interchange of atoms or groups between two or more compounds. For example, the reaction between lead nitrate (Pb(NO3)2) and potassium iodide (KI) to form lead iodide (PbI2) and potassium nitrate (KNO3) is a classic example of an exchange reaction.
What happens when a reaction reaches equilibrium?
When a reaction reaches equilibrium, the forward and backward reactions occur at the same rate. At this point, the concentrations of reactants and products remain constant over time. The equilibrium state does not mean that the reaction has stopped; rather, it signifies a dynamic balance between the forward and backward reactions.
Can you provide an example of a ligand exchange reaction?
Certainly! An example of a ligand exchange reaction is the reaction between silver nitrate (AgNO3) and sodium chloride (NaCl) in the presence of ammonia (NH3). In this reaction, the ammonia ligands replace the chloride ligands, resulting in the formation of silver chloride (AgCl) and sodium nitrate (NaNO3).
What is an ion exchange reaction example?
An ion exchange reaction involves the interchange of ions between two compounds. An example of an ion exchange reaction is the reaction between sodium chloride (NaCl) and silver nitrate (AgNO3) to form sodium nitrate (NaNO3) and silver chloride (AgCl). In this reaction, the sodium ions are exchanged with the silver ions.
Can you provide an example of a chemical exchange reaction?
Certainly! An example of a chemical exchange reaction is the reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH) to form sodium chloride (NaCl) and water (H2O). This reaction involves the exchange of hydrogen ions (H+) from the acid with the hydroxide ions (OH-) from the base.
Can you provide an example of an ion exchange reaction sequence?
Certainly! An example of an ion exchange reaction sequence is the reaction between calcium chloride (CaCl2) and sodium carbonate (Na2CO3) to form calcium carbonate (CaCO3) and sodium chloride (NaCl). In this reaction sequence, the calcium ions are exchanged with the sodium ions, resulting in the formation of the desired products.
Can you provide an example of a single exchange reaction?
Certainly! An example of a single exchange reaction is the reaction between zinc (Zn) and hydrochloric acid (HCl) to form zinc chloride (ZnCl2) and hydrogen gas (H2). In this reaction, the zinc atoms are exchanged with the hydrogen ions from the acid, resulting in the formation of the desired products.
Also Read: