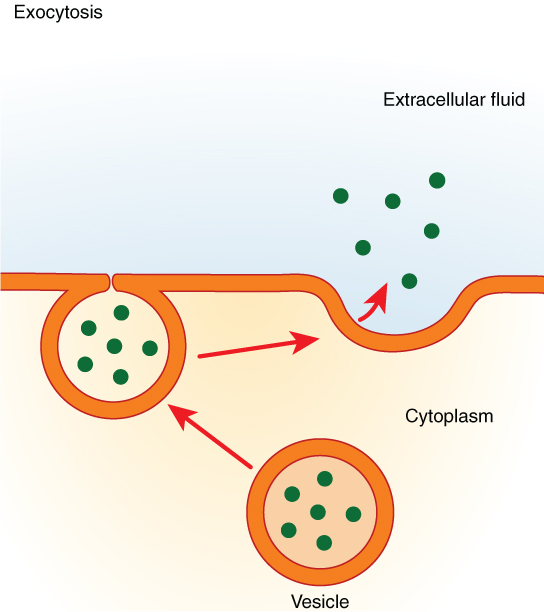
Exocytosis is the bulk transport of secretory molecules and toxins out of the cells involving the fusion of secretory vesicles with the plasma membrane.
Exocytosis also requires chemical energy in the form of ATP or GTP; thus, it is an active transport process. Endocytosis is the opposite of exocytosis in the sense that during endocytosis, material moves inside the cell instead of being moved out of the cell.
A small prerequisite point for the following discussion will be to remember that molecules are tagged with specific markers known as signal peptides. These signal peptides are responsible for either the residence of that molecule in a particular cell organelle or their movement through different subcellular compartments and subsequent packing into vesicles.
These cargo-containing vesicles can be stored for later use, or they can immediately move towards the plasma membrane to fuse with it and deliver their content to the extracellular compartment.
Types of molecules transported in exocytosis
Exocytosis involves transporting molecules like secretory proteins, waste materials, and neurotransmitters out of the cell. This process is also responsible for delivering lipids, carbohydrates and proteins to plasma membranes for growth and repair functions.
So, the type of molecules transported in this fashion range from proteins to carbohydrates to lipids and various other molecules.
What molecules move in exocytosis ?
Hormones, neurotransmitters, cellular junk material, neuropeptides, inflammatory molecules like histamines, proteins, lipids, proteoglycans and glycoproteins of the extracellular matrix make the list of chemicals that are exported out of the cell via exocytosis.
What happens in exocytosis ?
Cargo-containing vesicles fuse with the phospholipid bilayer. This fusion is mediated by various proteins that we will discuss shortly. Membrane proteins and the lipids of these vesicles provide new components for the cell’s plasma membrane; thus, the inner leaflet of these vesicles becomes the ectoplasmic surface of the lipid bilayer after the fusion.
Exocytosis can either be constitutive (continuous) or regulated exocytosis. The former is practically seen in all the cells. At the same time, the latter is found mainly in cells specialized for secreting products rapidly on demand—such as hormones, neurotransmitters, or digestive enzymes. Regulated depends upon calcium ions, while constitutive exocytosis does not.
Does exocytosis move from low to a high concentration?
This transport depends upon ATP, so the concentration is not exactly relevant. Another way of looking at it would be from the extracellular matrix (ECM) protein’s point of view.
The ECM already has a higher concentration of matrix proteins, but still, the cells can synthesize and deliver these proteins to the ECM, but all of this depends upon the requirement.
What does exocytosis use to move molecules?
Chemical energy in the form of ATP or GTP is used. Assembly of vesicles requires GTP and GTP-binding proteins. Apart from these, a microtubular network and various motor proteins are also needed to move these molecules from their site of synthesis to the target site of extrusion.
How does exocytosis work?
The general steps of exocytosis are vesicle trafficking, tethering, docking, priming (if needed), and fusing.
Vesicle trafficking-
Vesicle trafficking means the transport of vesicles to the plasma membrane. The material to be transported is packed into vesicles by the trans-Golgi network. These freshly pinched-off vesicles from the trans-Golgi network are known as immature vesicles.
Soon the similar immature vesicles fuse, and the material inside them becomes concentrated. These will be referred to as mature vesicles from now onwards.
Motor proteins and microtubules guide these vesicles to their site. Constitutive vesicles fuse with plasma membranes upon their arrival, but the regulated vesicles, like the ones containing hormones, neurotransmitters of enzymes, wait for the appropriate signal.
Tethering
Tethering involves contact between the vesicle and the plasma membrane.
Docking
Docking involves regular contact via proteins to the plasma membrane. Soon after docking, the membranes begin to fuse. Rab proteins (GTPases) are the key regulators of docking the vesicles onto the target membrane.
Rab-mediated docking is followed by the interaction of v-SNAREs with t-SNAREs to bring the membranes close enough to mediate the fusion. v-SNARE proteins on the vesicle join with t-SNARE proteins on the target membrane during the docking process.
The exact molecular details of fusion and its regulation are unknown.
After the membrane fusion, NSF and α-SNAP bind to the SNARE complexes. NSF hydrolyses the ATP to dissociated SNARE complexes, making them available for the next fusion round. Around the same time, GTP hydrolysis mediates the dissociation of Rab proteins from its effector molecules.
Priming
Priming involves modifications of some components required for proper fusion of membranes, and it only occurs in the case of regulated exocytosis.
Does exocytosis move water?
Yes, excess amounts of water are removed by exocytosis from the cell. Also, generally, the vesicles contain water-soluble proteins/cargo.
Hormone Arginine vasopressin mediates exocytosis of the water channel aquaporin 2 (AQP2) from the collecting duct cells, thus preventing excessive water loss.
Does exocytosis move through the phospholipid bilayer?
During exocytosis molecules movement does not occur through the phospholipid bilayer; rather, it involves fusion with the phospholipid bilayer. Every molecular detail of the fusion process is currently unknown, but it does involve the formation of SNARE complexes.
Does exocytosis move substances in vesicles?
Yes, exocytosis indeed involves vesicle formation. Specialized secretory cells store their secretions in secretory vesicles, also known as dense-core secretory granules. These secretory substances are selectively aggregated at the trans-Golgi network. However, the signals responsible for forming these aggregates are not well understood.
The uptake of these aggregates at the trans-Golgi network resembles the phagocytic uptake of particles at the cell surface. These vesicles become progressively concentrated with their content as they pass through the endoplasmic reticulum.
Progressive acidification of the vesicle lumen, retrieving excess membrane, and removing ER-resident proteins are some of the processes that immature vesicles go through to become dense core mature secretory vesicles.
Proteolytic cleavage of cargo:
Not only this, but cleavage of cargo to yield mature proteins is also one of the crucial steps during vesicle maturation. As most of the proteins, enzymes and hormones are synthesized in their inactive pre-form, they need proteolytic cleavage to achieve their mature functional forms. Sometimes the cleavage can also occur in the extracellular matrix after the exocytosis.
When exocytosis endocytosis occurs?
It depends upon the cellular requirement. If a cell needs to import many substances, then endocytosis is the pathway of choice, but for bulk export, a cell relies on exocytosis.
Examples of regulated exocytosis that lead to enlargement of the plasma membrane are cytokinesis, phagocytosis, wound repair and cellularization.
The cellularization process is seen in the fly embryo, which is initially a syncytium of nearly 6000 nuclei surrounded by a single plasma membrane. But within ten minutes, via regulated exocytosis, a vast number of cytoplasmic vesicles fuse and form plasma membranes around these nuclei, eventually forming 6000 cells.
Exocytosis transport direction
The direction of exocytosis is from the interior to the cell’s exterior.
Conclusion
The active bulk transport of materials from the cell is known as exocytosis, which could either be constitutive or regulated. Regulated pathways only function in some specialized secretory cells, while constitutive pathways are functional in all the eukaryotic cells.
Initially, immature vesicles bud off the trans golgi-network and mature into dense-core secretory granules stored for future use. Upon receiving an appropriate signal, these vesicles fuse with their target membrane to release their contents into the extracellular matrix.
Also Read:
- Plant cell parts and functions
- Why chromosomes are in pairs
- Ecosystem diagram
- Is diffusion passive
- Structure of cytosine
- Is coenzyme an enzyme
- Enzymes and respiratory
- Is a carbohydrate a monomer or polymer
- Purple sulfur bacteria photosynthesis
- Examples of extremophiles
Hi….I am Ashish Nandal, I have completed my Master’s in Biotechnology. I always like to explore new areas in the field of Biotechnology.
Apart from this, I like to read, travel, and photography.

Hi Fellow Reader,
We're a small team at Techiescience, working hard among the big players. If you like what you see, please share our content on social media. Your support makes a big difference. Thank you!